Abstract
Background: Ropeginterferon alfa-2b (Ropeg) is a novel long-acting monopegylated IFN-alpha-2b. Due to reduced dosing frequencies, better tolerability and improved compliance, Ropeg may be a favorable treatment option for long-term therapy in patients with polycythemia vera (PV).
Study design: PEGINVERA phase I/II (NCT: 2010-018768-18), a prospective, open-label, multicenter study, investigated the efficacy and safety of Ropeg for long-term treatment in 51 patients aged ≥18 years with a confirmed diagnosis of PV, regardless of prior cytoreductive therapy. Following ≥1 year of 2-weekly treatment, patients who responded well to Ropeg were permitted to switch to a 4-weekly dosing regimen.
Results: Baseline characteristics of the study cohort and interim safety and efficacy data were presented previously (Gisslinger et al., Blood, 2015). Fifty-one patients were treated: Median exposure to Ropeg was approximately 5.1 years (61 months; range: 0 to 87 months). Patients were treated for a median of approximately 2 years (98.9 weeks; (Q1-Q3: 69.0 - 117.4 weeks) on the 2-weekly regimen and 4 years (207.1 weeks; Q1-Q3: 158.6 - 242.0 weeks) on the 4-weekly regimen.
The best observed individual hematological response for patients in the efficacy analysis set (FAS) was a complete hematological response for 27/42 (64.3%) and a partial response for 14/42 (33.3%) patients. Patients required a median of 34 weeks (Q1-Q3: 10-96 weeks) treatment to achieve a complete hematological response, and 10 weeks (Q1-Q3: 10-20 weeks) to achieve any hematological response. Switch from 2 to 4-week dosing regimen had no apparent effect on maintenance of response.
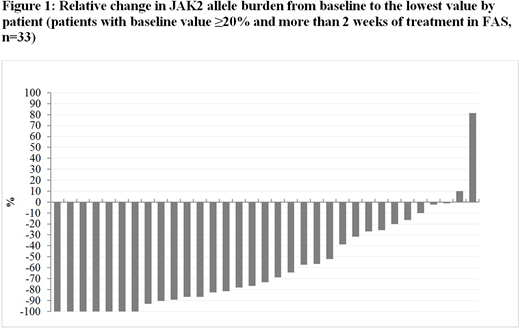
With respect to JAK-2 allele burden, the best observed individual molecular response was a complete response for 12/42 (28.6%) patients and a partial response for 19/42 (45.2%) patients. Lowest JAK-2 values relative to baseline are presented by patient in Figure 1. Patients required a median of 82 weeks (Q1-Q3: 44-115 weeks) treatment to achieve a complete molecular response and 34 weeks (Q1-Q3: 18-55 weeks) treatment to achieve any molecular response. Irrespective of dosing regimen, molecular responses tended to increase over time.
Most patients reported at least one adverse reaction (AR) to treatment (409 ARs in 48/51 [94.1%]); however, the majority (296 in 44 [86.3%] patients) were mild; 102 (in 34 [66.7%] patients) were moderate and 11 (in 10 [19.6%] patients) were severe. The most frequently reported ARs (frequency >20%) were arthralgia, influenza-like illness and fatigue. Twelve serious treatment emergent adverse events (TEAE) reported by 8/51 patients (15.7%) were considered to be treatment related: 2 events of depression, 2 of positive anti-thyroid antibodies, and one each of acute stress disorder, increased antinuclear antibodies, arthralgia, atrial fibrillation, fatigue, influenza-like illness, pyrexia, and increased transaminases.
25 patients completed the trial. The majority of discontinuation due to TEAE (13/21 patients) occurred in the first year, when the recommended slow up-titration of Ropeg could not be applied because of the maximum-tolerated-dose design. After the first year, only 8 additional patients discontinued because of TEAE.
Conclusions:
The final results of this phase I/II study of Ropeg in patients with PV support the findings of the pivotal phase III clinical trial (Gisslinger et al., Blood 2015) with respect to safety and efficacy as determined by hematological, clinical and molecular parameters. In addition, these data provide evidence that treatment with Ropeginterferon alfa-2b for up to 7 years is efficacious, well-tolerated and disease-modifying at both the 2 week and 4 week maintenance treatment regimens.
Gisslinger:AOP Orphan Pharmaceuticals AG: Consultancy, Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Janssen Cilag: Consultancy, Honoraria; Shire: Consultancy, Honoraria. Kralovics:MyeloPro Diagnostics and Research GmbH: Equity Ownership. Krauth:BMS: Consultancy, Honoraria; Celgene: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; Janssen: Consultancy, Honoraria. Greil:MSD: Honoraria, Research Funding; Merck: Honoraria, Research Funding; Sandoz: Honoraria, Research Funding; Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Astra Zeneca: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Honoraria, Other: TRAVEL, ACCOMMODATIONS, EXPENSES, Research Funding; Janssen: Other: TRAVEL, ACCOMMODATIONS, EXPENSES; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees. Zoerer:AOP Orphan Pharmaceuticals: Employment. Empson:AOP Orphan Pharmaceuticals: Employment. Grohmann-Izay:AOP Orphan Pharmaceuticals AG: Employment. Klade:AOP Orphan Pharmaceuticals AG: Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal